Overview of Mongolia’s Pharmaceutical Market: Insights from Delta Medical’s 15 years of Experience

However, Mongolia is difficult to access due to its remote location between Russia, Kazakhstan and China. Managing secure, cost-effective logistics is key for entering and remaining in the market. Delta Medical has 15 years of experience in Mongolia. The logistics remain challenging, but Delta Medical has experience managing secure pharmaceutical distribution solutions!
Mongolia Pharmaceutical Market Overview
Valued at $190 million as of 2021, Mongolia’s pharmaceutical market has witnessed steady expansion over the past decade, driven by economic development and investments in the healthcare system.
While still a nascent market compared to global standards, the country’s pharma sector is poised for rapid growth in line with the following key factors:
Expanding Healthcare Access and Infrastructure
- Healthcare expenditures as a share of GDP increased from 2.5% in 2017 to 3.5% in 2021
- Public health budgets expanded from $346 million in 2020 to $593 million in 2021
Rising Consumer Incomes and Health Awareness
- Mongolia’s nominal GDP has tripled over the past 10 years
- Citizens increasingly prioritizing healthcare and wellness
- Greater ability to pay for quality medicines and supplements
Favorable Population Trends
- Over 60% of population under age 35 indicating substantial future growth
- Rising rates of non-communicable diseases driving demand
- Expanding health insurance coverage currently at 95% of citizens
Growing Government Investments
- Substantial investments being made in primary care and hospitals
- New specialized oncology, cardiology and emergency hospitals constructed in Ulaanbaatar
- Medical tourism sector designated as a national priority – expected to bring more foreign revenue
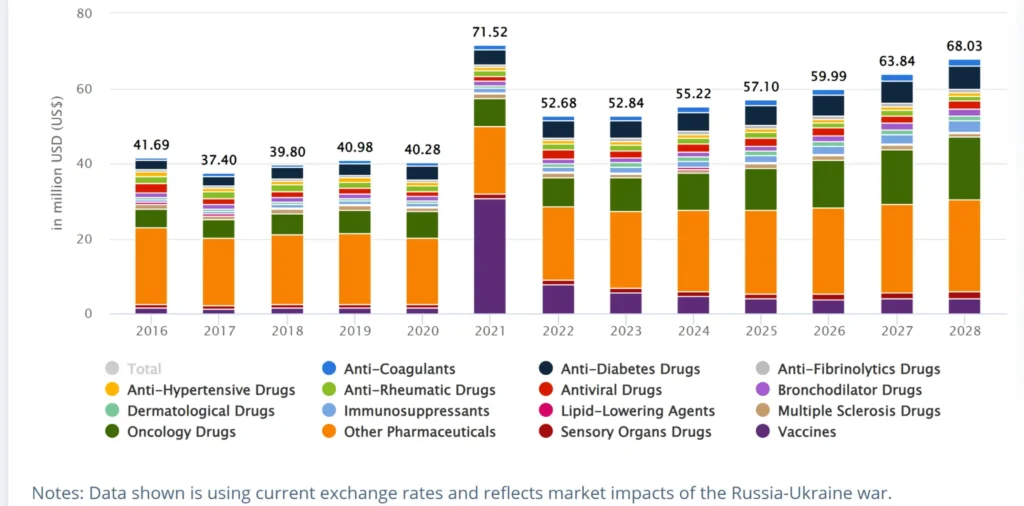
Table: Prescription Medicine Sales by Category in Mongolia
Pharmaceutical Market Segments
Mongolia’s pharmaceutical industry comprises the following key segments:
- Prescription Medicines: Largest segment, accounting for over 75% market share. Includes generic prescription pharmaceuticals as well as patented and branded medicines. Therapeutic areas with high usage include anti-infectives, respiratory, pain/inflammation, cardiovascular, and diabetes medicines.
- OTC Medicines: Over-the-counter pharmaceuticals make up around 20% of the overall market. Major OTC product categories are vitamins/minerals, digestive aids, pediatrics, dermatology, and pain relief. This segment is experiencing fast growth at 6% CAGR.
- Traditional & Herbal Products: Traditional Mongolian medicine and supplements have seen rising demand. Includes locally produced derivatives of medicinal plants, minerals and animal extract products. Projected to be an area of expansion.
- Medical Devices & Equipment: Currently limited market but expected to undergo substantial development. Orthopedics, patient monitoring, diagnostics and implantable device segments demonstrate high potential. Reliant on imports presently.
- Veterinary Medicines: Livestock sector is vital to Mongolia’s economy. Medicines and pharmaceuticals for animal health constitute a niche market segment.

Mongolia Pharma Regulatory Landscape
Mongolia’s pharmaceutical regulations and governance fall under the purview of the Ministry of Health and Sport (MOHS). Registration, quality assurance, licensing and other critical regulatory functions come under the mandate of divisions within the MOHS, including:
- State Registration Division for Medicines and Medical Devices
- Medicines Policy Implementation and Coordination Division
- Pharmaceuticals and Medical Devices Inspection Division
Key Regulations
The pharmaceutical sector adheres to stringent regulations outlined in the country’s Law on Medicines and Medical Devices,including:
- Registration protocols for drugs and medical equipment
- Quality assurance and standards compliance
- Licensing of manufacturers, importers and distributors
- Labeling and language requirements
- Adverse events monitoring
- Price controls for select essential medicines
- Prescriptions and dispensing protocols
Mongolia’s drug registration process typically takes between 12-18 months and requires specific dossier content as per national guidelines. Fast-track approvals are possible for products already registered in advanced markets like the U.S., EU, UK or Japan.
Importers must demonstrate a local infrastructure and capability to monitor quality and safety before bringing in pharmaceuticals.
Recent Regulatory Shifts
- The newly constituted Medicines and Medical Devices Regulatory Authority (MMDRA) as of December 2020 aims to consolidate regulatory functions, improve efficiency and align policies with international best practices.
- Stricter penalties have been introduced to curb illicit trading of falsified or substandard medicines given previous issues with black markets.
- New guidelines issued regarding pharmacovigilance obligations for companies to ensure adverse drug event monitoring and reporting capacity.
- An amended Patent Law now provides regulatory data protection and prohibits unfair commercial use of undisclosed test data for registered products.
Distribution Channels for Pharmaceuticals

Photo: Ulaanbaatar capital of Mongolia
Mongolia pharmaceutical products reach patients and consumers through hospitals, community pharmacies and a fast emerging online retail channel:
Hospital Pharmacies
- Around 43 hospital pharmacies currently operating in Mongolia attached to provincial, district, specialized and private hospitals
- Scope of services limited for most hospital pharmacies which act primarily as distributors of medicines within their respective hospitals
- Larger national hospitals in Ulaanbaatar have more advanced clinical pharmacy services including medication reviews, prescription analysis and rounding alongside physicians
Community and Retail Pharmacies
- Over 2300 privately owned pharmacy outlets as of 2021, concentrated in Ulaanbaatar city area
- Only licensed pharmacists can dispense medicines in these pharmacies
- Services mostly restricted to medication sales although some outlets provide telepharmacist consultations and home delivery
- New community pharmacy chains have expanded into the countryside improving access
Online Pharmacies
- Mongolia has over 60 online pharmacies catering to convenient medicine purchases
- Most e-pharmacies are extensions of existing brick-and-mortar outlets leveraging added digital presence
- Law on Medicines amended in 2019 to permit licensed online medicine retail under approved frameworks
- Projected 8.5% CAGR forecast for pharma ecommerce indicating shift in consumer preferences for digital access and delivery

Pharmaceutical Pricing, Reimbursement and Policy in Mongolia
The medicines pricing and reimbursement system aims to improve citizens’ access to affordable essential medicines. However, it faces limitations due to insufficient data and lack of transparency according to critics.
Price Controls and Policy
- Maximum retail prices (MRPs) imposed by Ministry of Health and Sport for medications included in the Essential Medicines List which is updated annually
- Price cap model sets acceptable mark-ups across supply chain stakeholders – manufacturers, importers, distributors, wholesalers and pharmacies
- Manufacturers must submit pricing analysis and get renewed approvals when applying for higher ceilings beyond threshold increases linked to inflation indices
Reimbursement and Subsidies
- National Health Insurance Fund covers majority of in-patient medicine costs across provincial hospitals and approved private facilities
- Out-patient reimbursements limited to select chronic diseases and a fixed annual budget allocation
- Critics argue current funding inadequately meets population needs citing shortages each month following exhaustion of earmarked budget
Ongoing Challenges
- Absence of pharmacoeconomic evaluation and lack of transparency in decision-making processes for setting medicine prices and reimbursement eligibility criteria
- High patient out-of-pocket expenditure at an average 60% of total medication costs due to narrow subsidization coverage
- Struggle to consistently enforce price controls especially in remote regions
Recent Policy Shifts
- Updated pricing regulations introduced mandatory cost analysis and government audits of pharmaceutical companies to ensure price ceiling compliance
- New Health Insurance Law aims to extend insurance coverage including out-patient prescription subsidies
- Government procurement centralized for select essential meds to improve availability by leveraging higher volumes
- Investments made into developing local pharmacoeconomics capacity to assist pricing determinations
Key Opportunities and Challenges in the Mongolia Pharma Sector
Mongolia presents an attractive emerging market for pharmaceutical companies but also one fraught with obstacles highlighting the dichotomy of opportunities and challenges:
Opportunities
- Government policy prioritizes local production indicating incentives for investments into domestic manufacturing
- Medical tourism and telemedicine sectors offer innovative partnership avenues
- Unmet clinical needs persist in areas like oncology, diabetes and cardiovascular diseases
- Scope for advanced digital health solutions to aid diagnosis, monitoring and enable remote consultations
Challenges
- Concerns over substandard drugs and previous black-market issues may discourage market entry unless strong vigilance systems implemented
- Current retail-based business models incur high operational costs in distributing across Mongolia’s vast geography and smaller population size
- Language barriers and availability of capable local talent
- Regulatory delays and administrative hurdles that extend time-to-market for registrants and applicants
“A man on the steppe with no friends is as narrow as a finger. A man with friends is as wide as the steppe.”
Mongolia presents an intriguing pharmaceutical market with ample opportunities for growth in coming years spurred by rising health expenditures and favorable demographic shifts.
However, companies seeking to enter this emerging landscape face considerable challenges navigating its regulatory standards, pricing policies, distribution complexities, and ethno cultural nuances. Success necessitates building strong local teams, infrastructure, and government relations able to manage Mongolia’s diverse market nuances.
As a leading pharmaceutical solutions provider expanding across Eurasia and CIS regions for over 20 years, Delta Medical offers specialized expertise for market entry into Mongolia.
Our comprehensive portfolio spanning regulatory support, product registration, supply chain logistics, commercialization, government relations and pharmacovigilance can catalyze your company’s launch plans.
We invite you to contact Delta Medical to discuss partnership opportunities for entering Mongolia. Our market-specific insights combined with your innovative pharmaceuticals and devices can improve patient access while exploring full growth prospects across this nascent but high-potential nation.

